The United Arab Emirates has been killing it lately; with the mission to Mars, and now a medicine to help treat COVID-19! The UAE, like many other countries, is facing the largest health crisis in modern history since the flu, and therefore, has created a medicine for the treatment of COVID-19. The UAE Ministry of Health and Prevention approved the emergency use of Sotrovimab, becoming the first country in the world to both license and enable immediate patient use!
The country’s efforts made to conduct COVID-19 tests and administer vaccines, along with the creation of this medicine puts the UAE among the world’s foremost countries that are effectively dealing with the COVID-19 pandemic. This new medicine will help with speeding up the recovery of patients and reducing hospitalization in intensive care units for more than 24 hours. Additionally, fatalities will be decreased by 85%.
Developed by GlaxoSmithKline (GSK) and Vir Biotechnology, the medicine is endorsed by the US Food and Drug Administration. More so, there are partnerships and agreements between the UAE health sector and international research institutions. Five studies have been done so far on Sotrovimab by the best institutions in the United States, the United Kingdoms, and South Africa. All of the research is dedicated to science and innovation.
One of the important aspects to pay attention to when handling this treatment is to ensure that the processes of licensing, shipments, distribution, and training for the doctors are all done professionally. Doctors need to be guided on how to use the medicines appropriately.

Sotrovimab
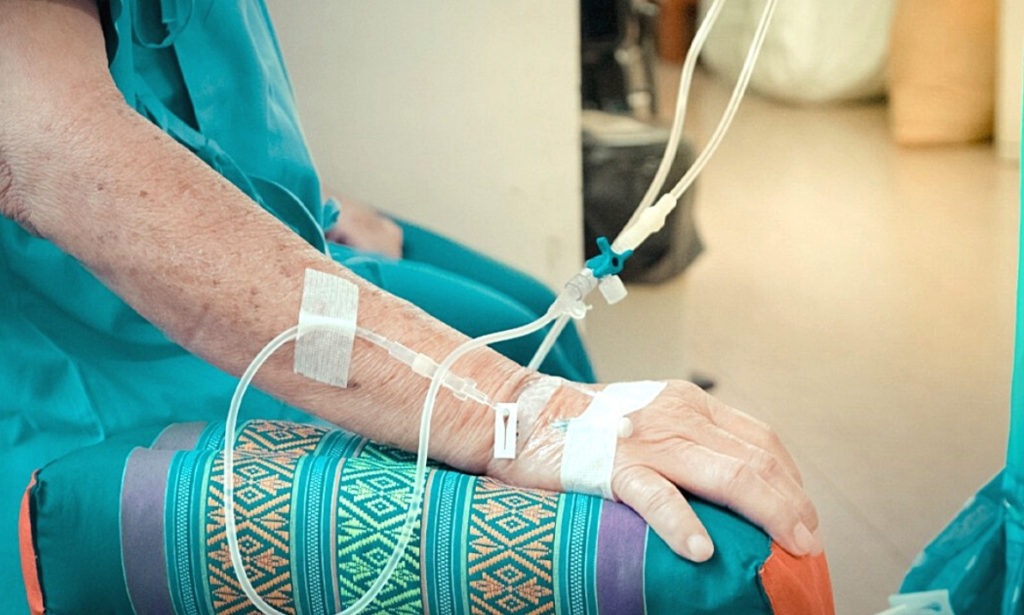
The treatment is meant for patients aged 12 years or older with mild to moderate COVID-19 symptoms; especially patients who are at risk of being hospitalized or are prone to death. Patients who are eligible to take this treatment need to weigh at least 40 kg. Sotrovimab is a single-dose monoclonal antibody. It’s a single IV infusion of 500 mg. It’s administered over 30 minutes and the patients should be monitored during the infusion and for at least an hour after the infusion.
Anyone with mild or moderate symptoms can take the treatment except patients who are hospitalized with COVID-19 or who require oxygen therapy for COVID-19, or who require an increase in baseline oxygen flow rate.